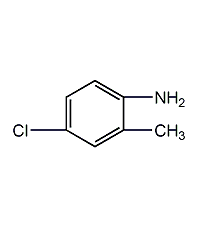
Structural formula
| Business number | 029L |
|---|---|
| Molecular formula | C7H8ClN |
| Molecular weight | 141.60 |
| label |
4-Chloro-o-toluidine, 2-amino-5-chlorotoluene, 2-Methyl-4-chloroaniline, 3-Chloro-6-aminotoluene, 4-Chloro-o-toluidine, 2-Amino-5-chlorotoluene, ClC6H3(CH3)NH2 |
Numbering system
CAS number:95-69-2
MDL number:MFCD00007842
EINECS number:202-441-6
RTECS number:XU5000000
BRN number:878505
PubChem number:24881294
Physical property data
1. Properties: flake crystal to brown liquid.
2. Density (g/mL, 20?): 1.190
3. Relative vapor density (g/mL, air=1): Undetermined
4. Melting point (ºC): 29~30
5. Boiling point (ºC, normal pressure): 241
6. Boiling point (ºC, kPa): Undetermined
7. Refractive index: 1.583
8. Flash point (ºC): 99
9. Specific rotation (º): Undetermined
10. Autoignition point or ignition temperature (ºC): Undetermined
11. Vapor pressure (mmHg, ºC): Undetermined
12. Saturated vapor pressure (kPa, ºC ): Undetermined
13. Heat of combustion (KJ/mol): Undetermined
14. Critical temperature (ºC): Undetermined
15. Critical Pressure (KPa): Undetermined
16. Log value of oil-water (octanol/water) distribution coefficient: Undetermined
17. Explosion upper limit (%, V/V): Undetermined
18. Lower explosion limit (%, V/V): Undetermined
19. Solubility: Undetermined
Toxicological data
1. Acute toxicity: rat oral LD50: 1058mg/kg; cat subcutaneous LDLo: 310mg/kg; wild bird oral LD50: 75mg/kg;
2. Mutagenicity
Mutation of microbial Salmonella typhimurium: 400?g/plate;
Mutation of microbial Salmonella typhimurium: 200?mol/plate;
Mutation of microbial Salmonella typhimurium: 250mg/ disc;
E. coli DNA repair: 2mg/disc;
Human HeLa cell mutation: 1mmol/L
Rat liver DNA synthesis: 100?mol/ L;
Oral DNA inhibition in mice: 200mg/kg;
Ecological data
This substance is harmful to the environment, and special attention should be paid to the pollution of water bodies.
Molecular structure data
1. Molar refractive index: 40.20
2. Molar volume (cm3/mol): 119.9
3. Isotonic specific volume??90.2K): 306.6
4. Surface tension (dyne/cm): 42.7
5. Polarizability (10-24cm3): 15.94
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 1
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 26
7. Number of heavy atoms: 9
8. Surface charge: 0
9. Complexity: 94.9
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
Avoid contact with strong oxidants, acids, acid anhydrides, and acid chlorides.
Storage method
Store in a cool, ventilated warehouse. Keep away from fire and heat sources. Protect from direct sunlight. Keep container tightly sealed. They should be stored separately from oxidants, acids, acid anhydrides and acid chlorides, and avoid mixed storage. Equipped with the appropriate variety and quantity of fire equipment. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
Originated from chlorination of o-toluidine. A variety of chlorination methods are available. o-Toluidine hydrochloride uses chlorobenzene as the solvent and reacts with chlorine at 50-60°C to obtain 4-chloro-o-toluidine. It can also be obtained by acylation first, then chlorination and hydrolysis. Mix and stir o-toluidine and chlorobenzene, add acetic anhydride dropwise, stir and react at 70-80°C for 1.5 hours, cool, and pass chlorine at about 50°C. At this time, a large amount of white solid will suddenly be generated. Stop passing chlorine, add concentrated hydrochloric acid and water, stir and reflux for 2 hours at 95°C. Cool to room temperature and neutralize with alkali solution. The yield is over 70%. Another method is to react o-toluidine hydrochloride with a gas mixture of hydrogen chloride and oxygen (or chlorine and oxygen) in an acidic medium.
Purpose
Used as dye intermediates.
extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/-PT303-tertiary-amine-catalyst–PT303-catalyst–PT303.pdfextended-reading:https://www.bdmaee.net/dimethyldecanoic-acid-dimethyl-tin-cas68928-76-7-dimethyldineodecanoatetin/extended-reading:https://www.bdmaee.net/dabco-rp202-catalyst-cas31506-44-2-evonik-germany/extended-reading:https://www.newtopchem.com/archives/940extended-reading:https://www.newtopchem.com/archives/44845extended-reading:https://www.bdmaee.net/cas-4394-85-8/extended-reading:https://www.newtopchem.com/archives/category/products/elastomer-catalystextended-reading:https://www.newtopchem.com/archives/category/products/page/170extended-reading:https://www.newtopchem.com/archives/1878extended-reading:https://www.bdmaee.net/fentacat-41-catalyst-cas112-03-5-solvay-2/

