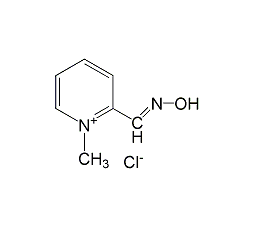
Structural formula
| Business number | 014G |
|---|---|
| Molecular formula | C7H9ClN2O |
| Molecular weight | 172.62 |
| label |
Pyridine-2-carboxaldehyde oxime methane chloride, pralidoxime chloride, pralidoxime chloride, 2-pyridine aldehyde, Phosphosphonate, Pyridine-2-carboxaldehyde oxime methyl chloride, Pyridine-2-carboxaldoxime methochloride |
Numbering system
CAS number:51-15-0
MDL number:MFCD00011981
EINECS number:200-080-9
RTECS number:UU4200000
BRN number:4163981
PubChem number:24899005
Physical property data
1. Properties: yellow-white crystalline powder.
2. Density (g/mL, 25/4?): Undetermined
3. Relative vapor density (g/mL, air=1): Undetermined
4. Melting point (ºC): 235-238? (decomposition)
5. Boiling point (ºC, normal pressure): Undetermined
6. Boiling point (ºC, normal pressure) 5.2kPa): Not determined
7. Refractive index: Not determined
8. Flash point (ºC): Not determined
9. Specific rotation ( º): Not determined
10. Autoignition point or ignition temperature (ºC): Not determined
11. Vapor pressure (kPa, 25ºC): Not determined
12. Saturated vapor pressure (kPa, 60ºC): Undetermined
13. Heat of combustion (KJ/mol): Undetermined
14. Critical temperature (ºC): Undetermined Determined
15. Critical pressure (KPa): Undetermined
16. Log value of oil-water (octanol/water) partition coefficient: Undetermined
17 . Explosion upper limit (%, V/V): Undetermined
18. Explosion lower limit (%, V/V): Undetermined
19. Solubility: in 25? water Solubility is 640mg/ml.
Toxicological data
1. Acute toxicity: human intravenous TDLo: 14mg/kg; human intravenous TDLo: 15mg/kg; rat intravenous LD50: 96mg/kg; rat intramuscular LD50: 150mg/kg; mouse oral LD50: 4100mg/kg; Mouse abdominal LD50: 155 mg/kg; Mouse intravenous LD50: 90mg/kg; Mouse intramuscular LD50: 100mg/kg; Dog intramuscular LD50: 75 mg/kg; Rabbit intravenous LD50: 95 mg/kg; Guinea pig intramuscular Internal LD50: 168mg/kg
Ecological data
None
Molecular structure data
1. Molar refractive index: not available
2. Molar volume (cm3/mol): not available
3. etc. Zhang Birong (90.2K):???Available
4. Surface tension (dyne/cm): None available
5. Polarizability (10-24cm3): None
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP):
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 3
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: 2
6. Topological molecular polar surface area (TPSA): 34.3
7. Number of heavy atoms: 11
8. Surface charge: 0
9. Complexity: 216
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters Number: 1
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 2
Properties and stability
None
Storage method
This product should be sealed and stored away from light.
Synthesis method
2-methylpyridine reacts with methyl chloride to obtain 2-methylpyridine methyl chloride salt. The base is nitrosated with ethyl nitrite to obtain pralidoxime chloride sodium salt, which is then dissolved in ethanol solution with concentrated hydrochloric acid. Mix to pH=3-4, filter out sodium chloride, and concentrate the filtrate under reduced pressure to recover ethanol to obtain crude pralidoxime chloride, which is dissolved in distilled water, decolorized with activated carbon, and recrystallized to obtain the finished product.
Purpose
2-methylpyridine reacts with methyl chloride to obtain 2-methylpyridine methyl chloride salt. The base is nitrosated with ethyl nitrite to obtain pralidoxime chloride sodium salt, which is then dissolved in ethanol solution with concentrated hydrochloric acid. Mix to pH=3-4, filter out sodium chloride, and concentrate the filtrate under reduced pressure to recover ethanol to obtain crude pralidoxime chloride, which is dissolved in distilled water; activated carbon is decolorized; and recrystallized to obtain the finished product.
extended-reading:https://www.newtopchem.com/archives/44254extended-reading:https://www.newtopchem.com/archives/39823extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/20-2.jpgextended-reading:https://www.bdmaee.net/high-efficiency-reactive-foaming-catalyst/extended-reading:https://www.newtopchem.com/archives/45191extended-reading:https://www.bdmaee.net/dimethylbis1-oxoneodecyloxystannane/extended-reading:https://www.bdmaee.net/foam-delay-catalyst/extended-reading:https://www.morpholine.org/bismuth-metal-carboxylate-catalyst-catalyst-dabco-mb20/extended-reading:https://www.bdmaee.net/pmdeta/extended-reading:https://www.bdmaee.net/wp-content/uploads/2021/05/2-10.jpg