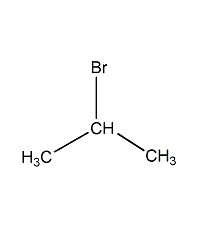
Structural formula
| Business number | 01JB |
|---|---|
| Molecular formula | C3H7Br |
| Molecular weight | 122 |
| label |
Aliphatic halogenated derivatives |
Numbering system
CAS number:75-26-3
MDL number:MFCD00000147
EINECS number:200-855-1
RTECS number:TX4111000
BRN number:741852
PubChem number:24892020
Physical property data
1. Properties: colorless or light yellow liquid[1]
2. Melting point (?): -89[2]
3. Boiling point (?): 59~60[3]
4. Relative density (water=1): 1.30[4]
5. Relative vapor density (air=1): 4.24[5]
6. Saturated vapor pressure (kPa): 23.275 (20?)[6]
7. Heat of combustion (kJ/mol): -2049.9[7]
8. Critical pressure (MPa): 5.51[8]
9. Octanol/water partition coefficient: 2.14[9]
10. Flash point (?): 1 (CC) [10]
11. Explosion limit (%): 7[11]
12. Lower explosion limit (%): 4[12]
13. Solubility: slightly soluble in water, soluble in ethanol and ether , benzene, carbon tetrachloride, slightly soluble in acetone. [13]
14. Heat of evaporation (KJ/kg, 58.6ºC): 231.07
15. Heat of fusion (KJ/kg, -89.0ºC ): 27.34
16. Heat of formation (KJ/mol): -125.2
17. Solubility parameter (J·cm-3)0.5: 17.068
18. van der Waals area (cm2·mol-1): 6.890×10 9
19. van der Waals volume (cm3·mol-1): 48.520
20 .Liquid phase standard claimed heat (enthalpy) (kJ·mol-1): -130.5
21. Liquid phase standard hot melt (J·mol-1·K-1): 142.2
22. Gas phase standard claims heat (enthalpy) (kJ·mol-1): -99.4
23. Gas phase standard entropy (J·mol-1·K-1): 316.13
24. Gas phase standard Free energy of formation (kJ·mol-1): -29.5
25. Gas phase standard hot melt (J·mol-1·K-1)?88.99
Toxicological data
1. Acute toxicity
Mouse abdominal LD50: 4837mg/kg;
Mammal inhalation LC50: 36 gm/m3;
2. Others Multiple dose toxicity data
Rat inhalation TCLO: 172 gm/m3/30M/1W-I;
3. Reproductive toxicity
Rat abdominal TDLO: 18900 mg/kgSEX/DURATION;
4.Acute toxicity[14] LC50: 31171ppm (mouse inhalation, 4h)
Ecological data
1. Ecotoxicity No information available
2. Biodegradability[15] MITI-I test, initial concentration 100ppm, sludge concentration 30ppm, 83% degradation after 4 weeks.
3. Non-biodegradability[16] In the air, when the hydroxyl radical concentration is 5.00×105 pieces/cm3, the degradation half-life is 18 days (theoretical). At 25°C, when the pH value is 7, the hydrolysis half-life is 2.1d.
Molecular structure number?
1. Molar refractive index: 23.63
2. Molar volume (cm3/mol): 91.6
3. Isotonic specific volume (90.2K ): 201.0
4. Surface tension (dyne/cm): 23.1
5. Polarizability (10-24cm3): 9.37
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 1.8
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 0
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 4
8. Surface charge: 0
9. Complexity: 10.8
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Stability[17] Stable
2. Incompatible substances[18] Strong oxidants, strong acids, strong bases
3. Conditions to avoid contact[19] Heating
4. Polymerization hazard[20] No polymerization
5. Decomposition products[21] Hydrogen bromide
Storage method
Storage Precautions[22] Store in a cool, ventilated warehouse. Keep away from fire and heat sources. The storage temperature should not exceed 37?. Keep container tightly sealed. They should be stored separately from oxidants, acids, and alkalis, and avoid mixed storage. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
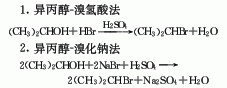
1. Obtained from the reaction of isopropyl alcohol and hydrobromic acid. Slowly add isopropyl alcohol to concentrated sulfuric acid under cooling, control the temperature below 30°C, add hydrobromic acid after the addition, slowly heat to reflux for 4 hours, and then distill until oil beads appear. The resulting crude product is divided into Wash with concentrated sulfuric acid, water and 5% sodium carbonate, then dry with anhydrous sodium carbonate, filter and fractionate. Collect the 58.5-60.5°C fraction as the finished product. There is also the isopropyl alcohol-sodium bromide method.
Purpose
Used in the synthesis of drugs, dyes and other organic compounds. [23]
extended-reading:https://www.bdmaee.net/nt-cat-dmaee-catalyst-cas1704-62-7-newtopchem/extended-reading:https://www.newtopchem.com/archives/44885extended-reading:https://www.bdmaee.net/cas-33568-99-9/extended-reading:https://www.newtopchem.com/archives/category/products/page/109extended-reading:https://www.bdmaee.net/dbu-octoate-polycat-sa102-niax-a-577/extended-reading:https://www.newtopchem.com/archives/40304extended-reading:https://www.cyclohexylamine.net/amine-catalyst-smp-delayed-catalyst-smp/extended-reading:https://www.newtopchem.com/archives/40292extended-reading:https://www.cyclohexylamine.net/dabco-amine-catalyst-amine-catalyst/extended-reading:https://www.newtopchem.com/archives/989

